


-
-
Scientific LibraryAcute Coronary Syndrom ASCVD Prevention Bifurcation Stenting Cardio-Oncology Congestive Heart Failure DAPT Duration Drug Coated Balloon Fractional Flow ReserveNewsCases VideosE-LearningIndustry Insights
- LIVE
-
 Article Link
Article Link

TAVI vs SAVR
CBSMD
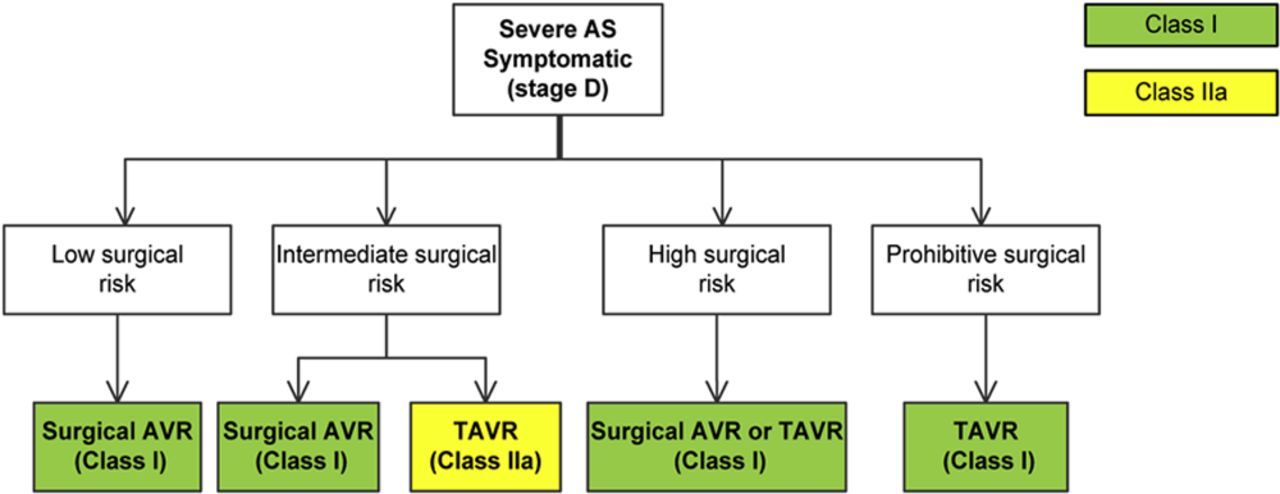
"Transcatheter aortic valve implantation (TAVI) is a class IIa recommendation for intermediate surgical risk and there is no recommendation for low surgical risk patients. Surgical aortic valve replacement (SAVR) is a class I for low, intermediate, and high surgical risk patients. Low-to-intermediate surgical risk patients comprised >90% of candidates for aortic valve replacement." Quoted from "Meta-Analysis of Effectiveness and Safety of Transcatheter Aortic Valve Implantation Versus Surgical Aortic Valve Replacement in Low-to-Intermediate Surgical Risk Cohort".
Class I of recommendation for SAVR treating low to intermediate risk patients and class IIa for TAVR treating intermediate risk patients with severe aortic stenosis which were given by "2017 AHA/ACC Focused Update of the 2014 AHA/ACC Guideline for the
Management of Patients With Valvular Heart Disease: A Report of the
American College of Cardiology/American Heart Association Task Force on
Clinical Practice Guidelines".

Figure 1. Choice of TAVR Versus Surgical AVR in the Patient With Severe Symptomatic AS.
"Contemporary real-world outcomes of surgical aortic valve replacement in 141,905 low-risk, intermediate-risk, and high-risk patients" demonstrated risk-stratified by Society of Thoracic Surgeons (STS) and in-hospital mean mortality in the most recent surgical era.
RCTs Comparing TAVI and SAVR
PARTENER 3 Trial - "Transcatheter Aortic-Valve Replacement with a Balloon-Expandable Valve in Low-Risk Patients"
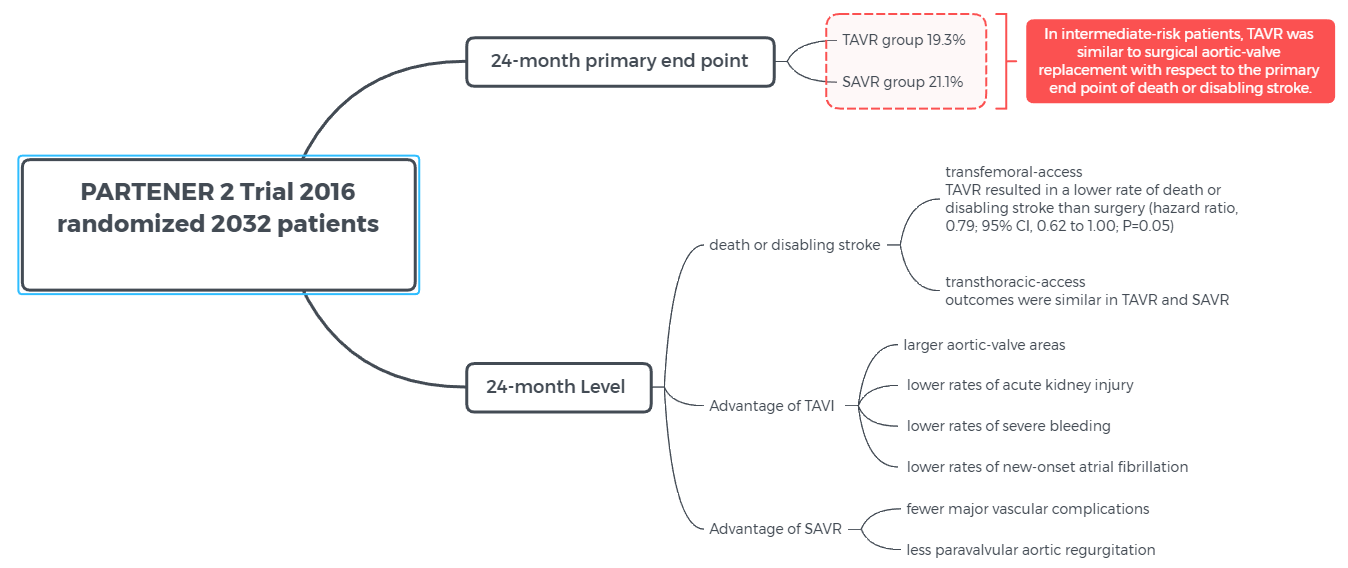
PARTENER 2 Trail - Among patients with aortic stenosis who were at intermediate surgical risk, there was no significant difference in the incidence of death or disabling stroke at 5 years after TAVR as compared with surgical aortic-valve replacement.
2-year follow-up outcome published in 2016 : "Transcatheter or Surgical Aortic-Valve Replacement in Intermediate-Risk Patients"
5-year follow-up outcome published in 2020: "Five-Year Outcomes of Transcatheter or Surgical Aortic-Valve Replacement"
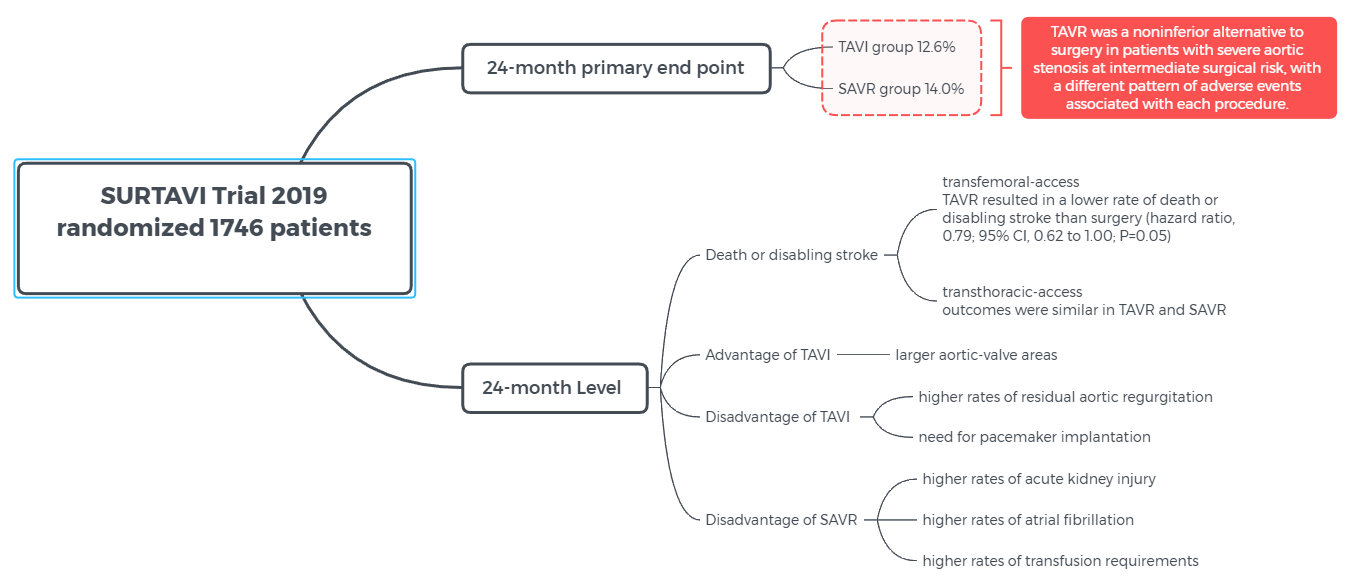
SURTAVI Trial - "Surgical or Transcatheter Aortic-Valve Replacement in Intermediate-Risk Patients"
Please click on the article link on top right to get further access to the CoreValve Clinical Trail, STACCATO Trail and NOTION Trial.
New Evidences Presented at ACC 2020
PARTNER 3 trial 2 Year Follow-up - TAVR is Equivalent to Surgery at Two Years in Low-Risk Patients

Michael Mack, M.D., presenting the primary endpoint slide of combined death, stroke or rehospitalization in TAVR vs. surgery patients in the PARTNER 3 trial presented at ACC 2020.

The two-year outcomes also indicate patients undergoing TAVR had a significantly higher rate of valve thrombosis, the formation of blood clots on the valve, which occurred in 2.7 percent of patients in the TAVR group and 0.7 percent of those who received surgery at two years. However, there was no significant deterioration in the functioning of the valve itself between years one and two in either study group.
UK TAVI - Compared TAVR vs SAVR in Patients with Severe Aortic Stenosis

Conclusions from the real world TAVR vs. surgery trial for aortic valve
replacement outcomes in the U.K., presented by William D. Toff, M.D. at
ACC 20.
The trial enrolled 913 patients referred for treatment of severe aortic stenosis at 34 U.K. sites from 2014-2018. All types of valves were included. Half were randomly assigned to receive TAVR and half underwent SAVR. Enrollment was limited to participants age 70 years or older (with additional risk factors) or age 80 years or older (with or without additional risk factors). Overall, participants were at intermediate to low risk from surgery, with a median Society of Thoracic Surgeons (STS) risk score of 2.6% (a score below 3% is considered low risk). At one year, the rate of death from any cause was 4.6% among patients undergoing TAVR and 6.6% among those undergoing open-heart surgery, a difference that met the trial’s pre-specified threshold for non-inferiority of TAVR. Rates of death from cardiovascular disease, stroke and a composite of those two outcomes were similar between the two groups.
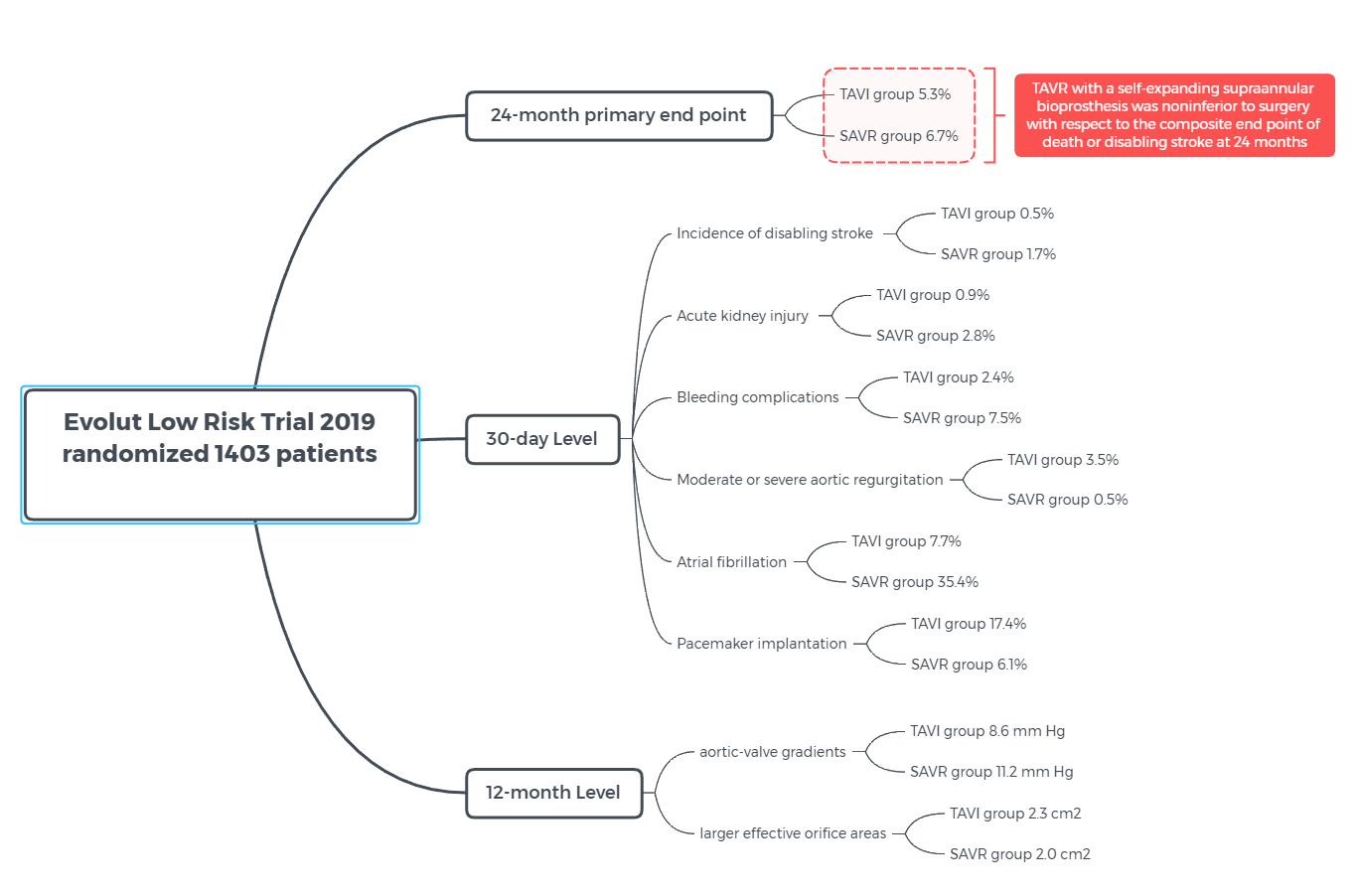
Evolut - Low Risk Bicuspid Study - Transcatheter Aortic Valve Replacement with Severe Bicuspid Aortic Confirms Transcatheter Procedure is a Viable Alternative to Open Heart Surgery

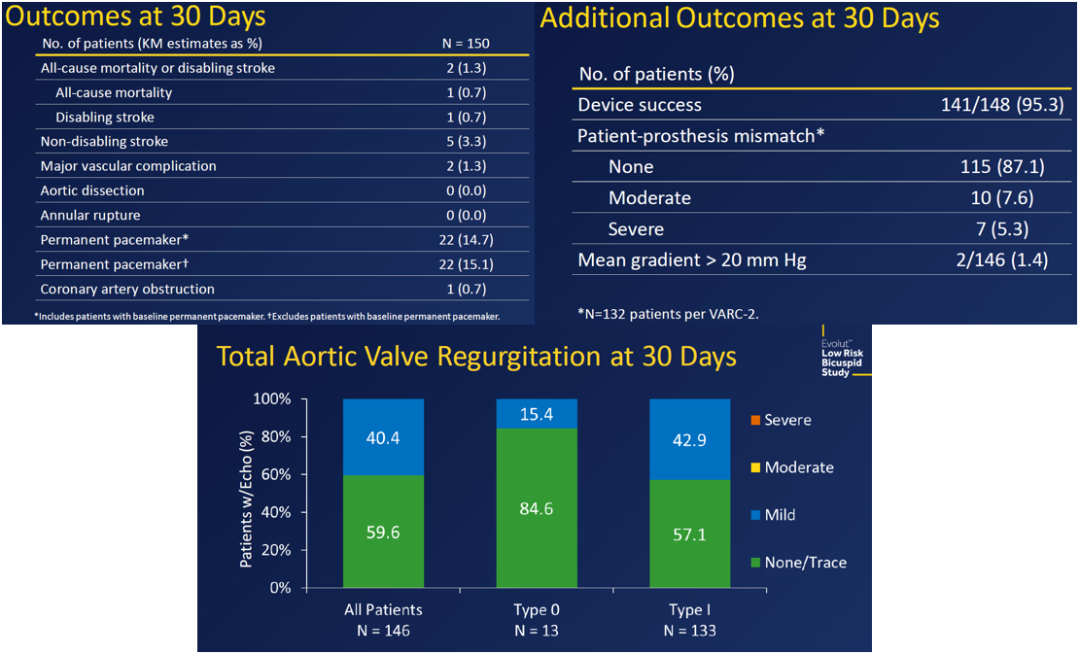
The study prospectively tracked 150 patients who underwent TAVR at 25 medical centers in the U.S. On average, patients were 70 years old and had a Society of Thoracic Surgeons (39) risk score (which reflects the risk of dying during or shortly after open heart surgery) of 1.4 percent.
Thirty days after their procedure, just 1.3 percent of patients had died or experienced a disabling stroke, the study’s primary safety endpoint. Patients also had an overall device success rate of 95.3 percent, reflecting a low rate of major complications, ill-fitting valves or the need for additional procedures. The vast majority (99.3 percent) of patients survived the procedure, 96 percent showed correct positioning of the valve and 100 percent had mild or no aortic regurgitation (blood leaking around the valve). This is the first study that shows the self-expanding valve works well for patients with both type 1 and type 0 bicuspid valves.

http://www.cbsmd.cn Contact us by cbs@cbsmd.cn
Copyright ⓒ CBSMD Nanjing China. All rights reserved.



